Budget ignores life-changing new and future medicines despite global challenges
12 May 2026: Medicines Australia is gravely concerned that the 2026–27 Federal Budget has not included a funded plan to implement long-awaited improvements to patient and consumer access to new and innovative medicines, and a key pathway for these reforms – Australia’s Health Technology Assessment (HTA) system.
Long-held fears over the vitality and speed of the Pharmaceutical Benefits Scheme (PBS) have been deepened by growing international uncertainty created by the United States’ proposed Most Favoured Nations (MFN) pricing policy. Without a timely, clear and coordinated response, there is a real risk Australian patients and consumers become victims of a less attractive market for the development and launch of new and innovative medicines and therapies.
While the PBS remains one of Australia’s most important public health programs, the system used to assess and list medicines has not undergone substantial reform in more than 30 years. As a result, Australian patients are increasingly waiting longer for access to innovative treatments that are available in comparable countries – or missing out entirely.
Despite years of reviews, expert consultation and consensus-driven recommendations, the Budget contains no meaningful funding commitment or progress on implementation of HTA reform. Failure to further invest – at a time other countries are increasing their funding of new medicines – will ultimately come at a cost to patients and consumers.
While Medicines Australia acknowledges the headline $5.9 billion (on which the Government will be rebated billions from industry) to cover recently listed PBS medicines, the reality is that the 2026-27 Budget shows that ongoing investment into the PBS – and health generally – over the forward years is set to decline as a proportion of Gross Domestic Product (GDP).
Without an increase in PBS spending as a proportion of GDP, system challenges will remain in ensuring access to new and innovative medicines by Australian patients.
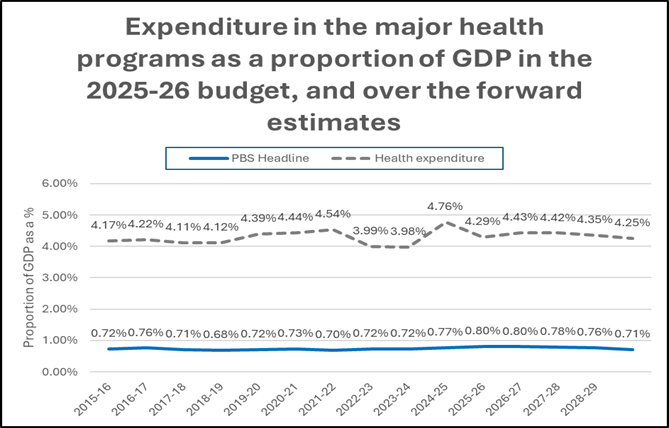
(Source: Budget Paper 1; MA analysis)
Medicines Australia understands that with the ongoing cost-of-living challenge that Australian families face that efforts to keep the price of medicines down are a priority, the fact remains that without further reforms to the HTA, Australians will simply struggle to access the new and innovative medicines that will make a difference to their health.
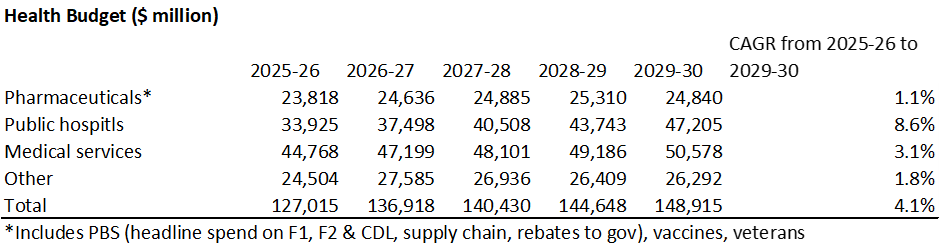
(Source: Budget Paper 1)
We urge the government to prioritise the accessibility and availability of new medicines at the forefront of reforms to the PBS rather than simply treading water.
As part of Budget 2026-27, we welcome the creation of a National Science and Resilience Council but emphasise the need for a medicines industry representative to be included in the Health working group.
Increased spending through the Medical Research Future Fund is welcomed along with the contributions to the National One Stop Shop and a not-for-profit clinical trials program. But they are no substitute for the broad funding and structural reform the PBS so badly needs.
Medicines Australia remains committed to working constructively with the Australian Government and Health Minister Mark Butler to modernise Australia’s medicines system and ensure patients have timely access to the treatments they need. However, there are deepening concerns about the pace and priority being given to improving patient access to innovative medicines in an uncertain world.
From 2014-2019, there was, on average, a delay of 391 days between the TGA deeming a new medicine as safe and effective and Australians receiving subsidised access to it. From 2016-2021, this delay increased by 19 per cent, jumping up to an average of 466 days.
Quotes attributable to the Chief Executive Officer of Medicines Australia, Liz de Somer:
“Medicines Australia understands that Australia is facing a number of substantial economic challenges but there should always be room in the federal Budget to improve access for patients to new and innovative medicines.”
“Australia should be proud of the PBS and the role it plays in ensuring affordable access to medicines, but the system supporting it has not kept pace with advances in medical innovation.”
“Patients are waiting too long for access to new treatments, and without meaningful reform to the HTA system we risk falling further behind comparable countries.”
“The emergence of international pricing policies such as the United States’ Most Favoured Nations proposal creates additional uncertainty for global medicine investment and launches.”
“Australia cannot assume it will automatically remain an early access market for innovative medicines. Maintaining that position requires a modern, efficient and globally competitive system.”
“We stand ready to continue working constructively with Government to ensure Australians can access the medicines and therapies they need, when they need them.”
ENDS
BACKGROUND ONLY
Reform of the HTA to ensure medicines are listed on the PBS – it’s time to take action.
It has been five years since the 2021 New Frontiers report the growing concerns with Australia’s PBS including outdated methods, policies and processes, and highlighted that patients are waiting too long to access new, innovative and life changing treatments.
The 2023, the HTA Policy and Methods Review reiterated these issues and developed 50, consensus driven, recommendations derived from hundreds of submissions from thousands of patients and clinicians, as well as industry and government stakeholders.
The 2024 Productivity Commission report Advances in measuring healthcare productivity states that Australia gets relatively good and increasing value for its healthcare dollar, putting productivity gains at around 3 per cent.
The 2024, independently chaired Implementation Advisory Group spent another year developing a Roadmap to assist the Government to implement the HTA Review recommendations, which was delivered in 2025.
Then recently, the Department of Health has engaged in another round of unnecessary consultation, without seeking advice from the industry through Medicines Australia. While consultation is critical, it is just as important that extra hurdles are not created as to prevent action being taken quickly or using extra consultation as a barrier to implementing the already-agreed to HTA reforms.
Ahead of the 2026 Budget, the Medicines Australia Pre-Budget Submission made four recommendations to Government for consideration to help drive greater access to new medicines:
- Increase investment in innovative medicines and implement HTA reform
- Focus on reforming lowest cost comparator and discount rate applied to appropriately value innovative medicines
- Continue to drive NOSS to boost research ecosystem for innovative medicines
- Establish a national life sciences council
Medicines Australia Pre-Budget Submission: https://www.medicinesaustralia.com.au/wp-content/uploads/sites/65/2026/02/20260130_Medicines-Australia_Pre-Budget-Submission-2026-27.pdf
How spending on the PBS is accounted for in the federal Budget:
Spending on the Pharmaceutical Benefits Scheme (PBS) is accounted for in the federal Budget as a demand-driven expense, with costs based on the volume of subsidised prescriptions and listing of new medicines.
PBS expenditure is reported on an accrual basis, reflecting the cost of medicines supplied in the financial year.
PBS expenditure is not explicitly capped, meaning spending can rise with increased demand and the addition of new, often high-cost, drugs, although the government projects these costs as a steady percentage of GDP.
PBS expenditure is reported in MYEFO and the annual federal Budget.
It is reported through Department of Health and Aged Care Portfolio Budget Statements
Australians are missing out
Only around 27 percent of globally supplied innovative medicines are made available to Australians via the PBS.
Media enquiries to:
Jonathan Hawkes, Senior Media Advisor, Medicines Australia – 0434 660 801 or Jonathan.hawkes@medicinesaustralia.com.au
Anne-Marie Sparrow, Medicines Australia /Cube – media@medicinesaustralia.com.au or 0417 421 560.
ABOUT MEDICINES AUSTRALIA
Medicines Australia leads the research-based medicines industry of Australia. Our members discover, develop and manufacture prescription pharmaceutical products, biotherapeutic products and vaccines that bring health, social and economic benefits to Australia. Our members invest in Australian medical research and take local discoveries and developments to the world.
